In this week’s News of Note, Amazon continues its competition with Starlink by launching another batch of internet satellites, WhatsApp receives a ban by congressional staffers and “the ChatGPT of quantum computing” launches in Canada. Elsewhere, Texas Instruments announces a major investment in semiconductor production in the United States.
Articles Posted in Cannabis
Cannabis Reform in 2021: New Era of Flux?
 With the inauguration of President Joseph Biden in January and the attaining of a narrow majority by Democratic party in Congress, there’s been some excitement in the cannabis space, as well as many questions revolving around one central unknown: How will the Biden administration and the swing in Congress leadership impact cannabis reform?
With the inauguration of President Joseph Biden in January and the attaining of a narrow majority by Democratic party in Congress, there’s been some excitement in the cannabis space, as well as many questions revolving around one central unknown: How will the Biden administration and the swing in Congress leadership impact cannabis reform?
COVID-19 and Cannabis: The Pandemic Complicates the Prospects of an Already Complicated Industry
 While everyone is chilling at home, well into over a month of mandatory social distancing, demand for cannabis products has never been higher. Sales of cannabis products broke records last months, helped by the transition of many stores to delivery or curbside pick-up. Sales have spiked in California, Colorado, Nevada, Oregon and Washington—all of which are states in which recreational usage of marijuana was legalized. Cannabis-related beverages and edibles have especially seen an uptick in interest, with edibles being estimated to have an impressive 28% increase in growth. Customers in some states have also been observed to be panic-shopping cannabis products in bulk, along with their stockpiles of toilet paper, yeast, alcohol and other groceries, indicating the increasing importance of cannabis-related products as everyday consumer products.
While everyone is chilling at home, well into over a month of mandatory social distancing, demand for cannabis products has never been higher. Sales of cannabis products broke records last months, helped by the transition of many stores to delivery or curbside pick-up. Sales have spiked in California, Colorado, Nevada, Oregon and Washington—all of which are states in which recreational usage of marijuana was legalized. Cannabis-related beverages and edibles have especially seen an uptick in interest, with edibles being estimated to have an impressive 28% increase in growth. Customers in some states have also been observed to be panic-shopping cannabis products in bulk, along with their stockpiles of toilet paper, yeast, alcohol and other groceries, indicating the increasing importance of cannabis-related products as everyday consumer products.
FDA Public Hearing on Regulation of CBD: Are Extract, Processing and Quality Control Technologies Potentially More Important than Ever in the Cannabis Industry?
 On May 31, 2019 the FDA held a public hearing about cannabidiol (CBD) products. The day-long hearing saw comments and presentations from stakeholders ranging from nonprofit organizations like ASTM International to the Grocery Manufacturers Association, a food, beverage and consumer product trade association. Corporate entities in the cannabis industry also provided testimony, such as ingredient manufacturer Mile High Labs, which supplies products like CBD isolate and concentrate, and Socati, which provides high-CBD genetic varieties and extraction processes. Pharmaceutical companies like Zynerba Pharmaceuticals Inc., which produces the drug Connect-FX, were also present. Connect-FX is a CBD transdermal gel for treating Fragile X syndrome (Martin-Bell syndrome), a rare genetic disorder that causes developmental problems like intellectual disabilities. It is currently being evaluated as an experimental treatment for use in child patients in clinical trials.
On May 31, 2019 the FDA held a public hearing about cannabidiol (CBD) products. The day-long hearing saw comments and presentations from stakeholders ranging from nonprofit organizations like ASTM International to the Grocery Manufacturers Association, a food, beverage and consumer product trade association. Corporate entities in the cannabis industry also provided testimony, such as ingredient manufacturer Mile High Labs, which supplies products like CBD isolate and concentrate, and Socati, which provides high-CBD genetic varieties and extraction processes. Pharmaceutical companies like Zynerba Pharmaceuticals Inc., which produces the drug Connect-FX, were also present. Connect-FX is a CBD transdermal gel for treating Fragile X syndrome (Martin-Bell syndrome), a rare genetic disorder that causes developmental problems like intellectual disabilities. It is currently being evaluated as an experimental treatment for use in child patients in clinical trials.
Cannabis, Yeast and the Quest for Cannabinoid Synthesis
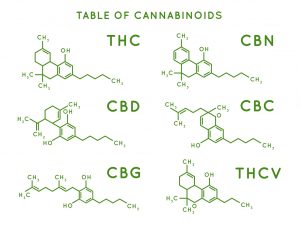 CBD, CBG, CBA, CBN, THC—the race to find the holy grail of cannabinoid production is in full swing. Money flows abound, unicorn-hungry investors looking to capture market share are swirling around promising frontrunners with lucrative IP. One interesting segment of cannabis IP gaining traction focuses on cannabinoids synthesis from microorganisms such as yeast.
CBD, CBG, CBA, CBN, THC—the race to find the holy grail of cannabinoid production is in full swing. Money flows abound, unicorn-hungry investors looking to capture market share are swirling around promising frontrunners with lucrative IP. One interesting segment of cannabis IP gaining traction focuses on cannabinoids synthesis from microorganisms such as yeast.
Going for the Green, Part II: Patents for Cannabis Plant Processing Methods, Coffee Pods and Condoms
 Given the growth of investments in and shift of regulatory views regarding cannabis-related products, many companies in industries like medicine, lifestyle and foods/beverages are looking to carve out niches and be leaders in the relatively new space. As with any new technology space, it is essential to have a robust intellectual property protection strategy to both establish and preserve one’s position as a dominant player in an emerging market. One important step that a company may take when creating such a strategy is applying for patents.
Given the growth of investments in and shift of regulatory views regarding cannabis-related products, many companies in industries like medicine, lifestyle and foods/beverages are looking to carve out niches and be leaders in the relatively new space. As with any new technology space, it is essential to have a robust intellectual property protection strategy to both establish and preserve one’s position as a dominant player in an emerging market. One important step that a company may take when creating such a strategy is applying for patents.
Going for the Green, Part I: Epilepsy Drugs, Sparkling Water and the Continued Shift in Government Regulation Concerning Cannabis-Based Products
 Over the past several years, cannabis has been one of the hottest areas of investment and innovation, with many states introducing legislation to legalize cannabis use in some form. Correspondingly, many companies have entered the U.S. market and are even listed on the Nasdaq or the New York Stock Exchange, leading to much interest on Wall Street. Many nascent industries have budded in the cannabis space, ranging from growing the cannabis plant itself to extraction processes to consumer products like vapes.
Over the past several years, cannabis has been one of the hottest areas of investment and innovation, with many states introducing legislation to legalize cannabis use in some form. Correspondingly, many companies have entered the U.S. market and are even listed on the Nasdaq or the New York Stock Exchange, leading to much interest on Wall Street. Many nascent industries have budded in the cannabis space, ranging from growing the cannabis plant itself to extraction processes to consumer products like vapes.
 Internet & Social Media Law Blog
Internet & Social Media Law Blog